The Food and Drug Administration issued a warning after a social media post by Kim Kardashian sparked concerns about the way information on drugs is being disseminated, The New York Times reports.
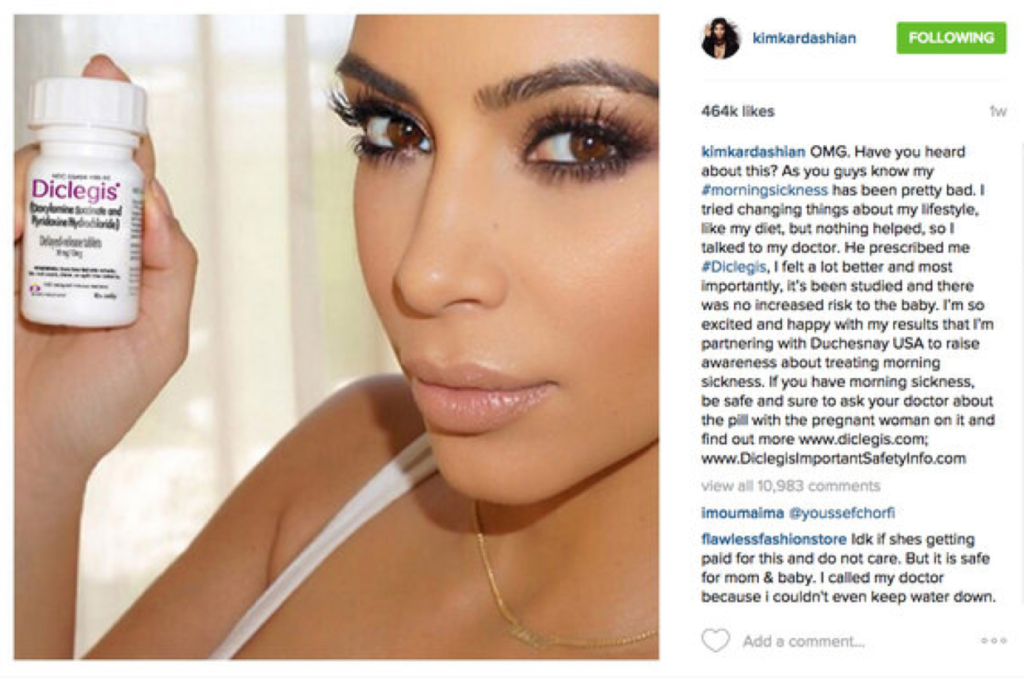
Kardashian, who is pregnant and has been battling morning sickness, posted on Instagram about the prescription drug Diclegis, which she said worked so well that it convinced her to partner with the drug’s manufacturer, Duchesnay, to get the word out about the drug.
“The Food and Drug Administration struck back against Ms. Kardashian’s post, issuing a warning letter (which it made public this week) to the drug’s maker, Duchesnay Inc., that said the post failed to give information about the drug’s risks,” The Times reports. “The letter ordered the company to ‘immediately cease misbranding.’”
The report adds: “It was the latest among several actions the agency has taken against posts on sites like Facebook and YouTube. Thomas Abrams, the director of the drug promotion office, said in a 2013 interview that social media had complicated the agency’s ability to monitor drug promotions, citing the sheer volume of content online and the speed with which it can be changed.”

What a stupid pig! This person has used up her “15 minutes” of fame many years ago. The big-ass bimbo should be boycotted by all right-thinking people…though her slavish, sleazy public will no doubt continue to watch her mind-numbing show and silly activities and inane comments.